CHANNELS

Group Leader
Prof. Dr. med. Bernhard Schermer
+49 221 478-89030
bernhard.schermer@uk-koeln.de
TEAM MEMBERS
Martyna Brütting
Torsten Bücher
Inês Cabrita, PhD
Lena Ebert, PhD
Lioba Ester
Emilia Kieckhöfer
Xinyi Lin
Gisela Slaats, PhD
Bernhard Schermer
Cilia Biology and Cystic Kidney Disease
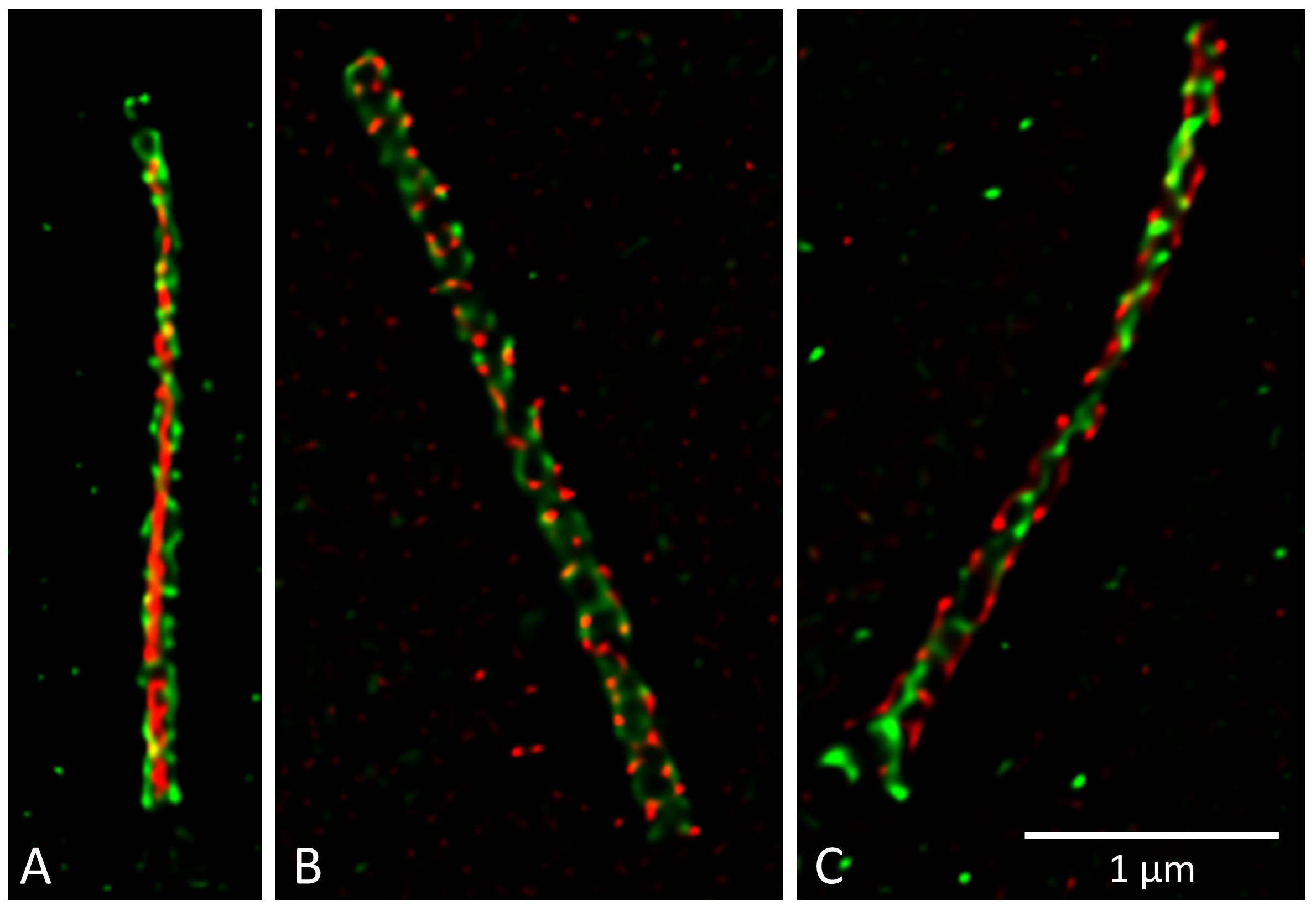
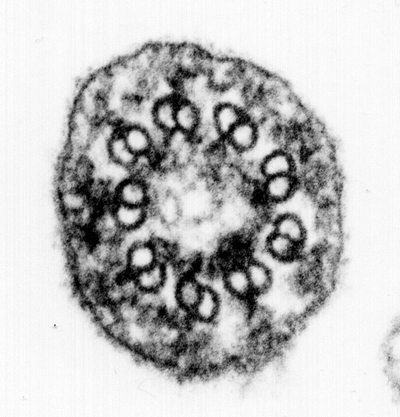
Hereditary cystic kidney diseases such as autosomal-dominant polycystic kidney disease (ADPKD) or nephronophthisis (NPH) are among the most common causes of end-stage renal failure worldwide. These diseases have been linked to mutations in genes encoding for proteins that localize to a specialized compartment of tubular renal epithelial cells, the primary cilium. Primary cilia are sensory organelles that project from the surface of virtually all cells of our body. A cilium is covered by a specialized plasma membrane and consist of the ciliary axoneme and the basal body, a modified centriole.
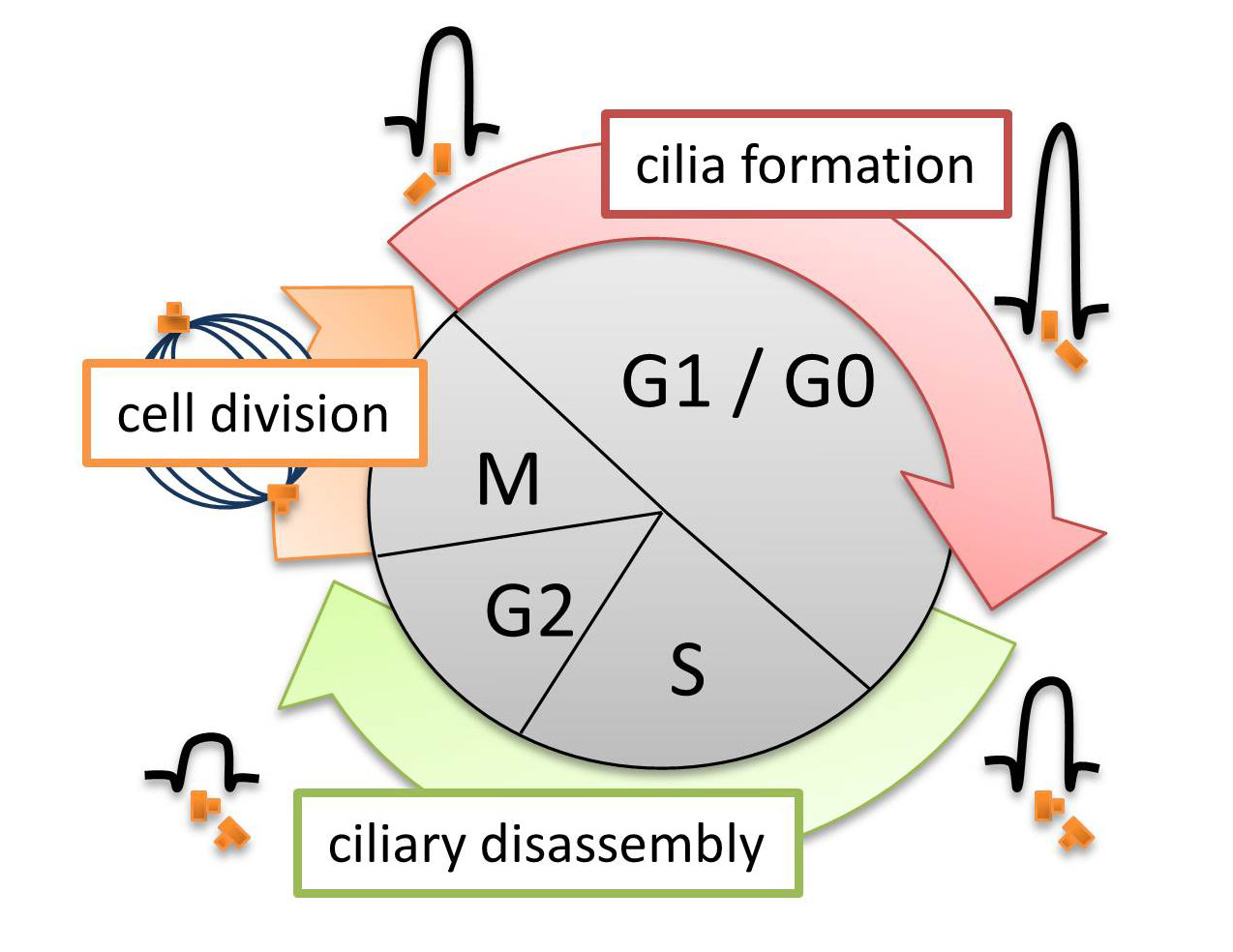
Cilia have been shown to act as antennae that receive and transmit signals from the cell’s environment; the remote information is then converted into signaling cascades that are initiated within the ciliary compartment and transmitted into the cell. Cilia are mainly formed by quiescent cells in interphase and are thus temporally associated with the development of cell polarity and differentiation. Cilia typically disassemble when the cell re-enters the cell cycle prior to mitosis.
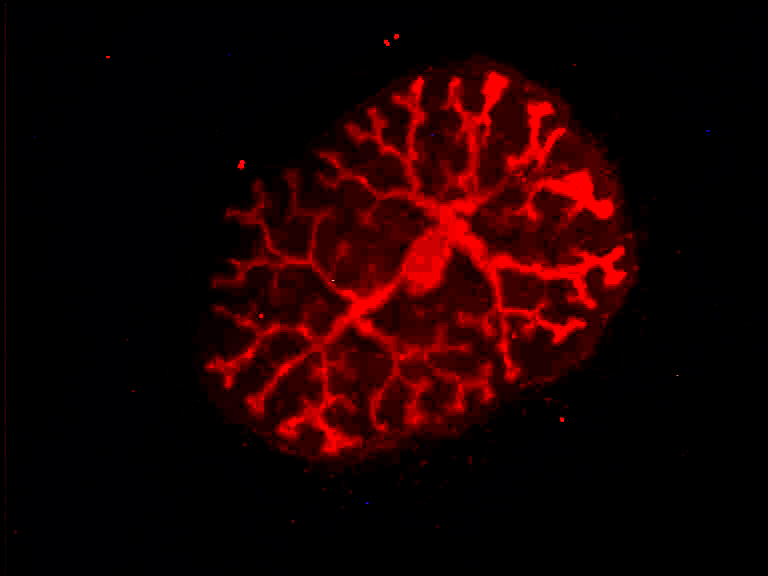
Members of our team are working on various aspects of ciliary function, focussing on genetic cystic kidney disease (ADPKD, NPH, NPH-related ciliopathies). Our group was the first to show that proteins involved in the development of nephronophthisis (NPH) are present in cilia of kidney cells. We could further demonstrate that these NPH proteins form multiprotein complexes that are involved in signaling from cilia to the interior of the cell. Moreover, we studied how NPH proteins modulate both Wnt and Hippo signaling.
Currently, we are working on the identification of novel ciliary protein complexes using proteomics technologies, and we are addressing the in vivo relevance and function of protein interactions using the mouse and C. elegans as model systems combined with various genome engineering approaches based on CRISPR/Cas9. To this end, we aim to understand the signaling networks involved in the pathogenesis of cilia-associated diseases. Ongoing projects aim to understand the role of Jade-protein family members in ciliopathies, to decipher the function of RNA binding proteins in cilia biology, to investigate regulated cell death in the context of ciliopathies, and to test novel therapeutic approaches in preclinical models of NPH.
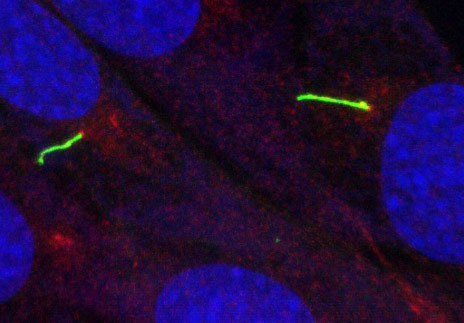
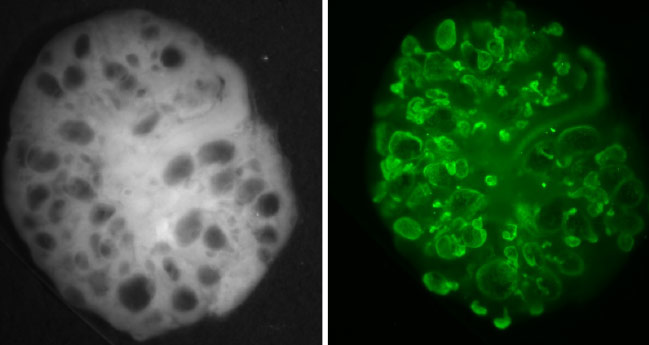
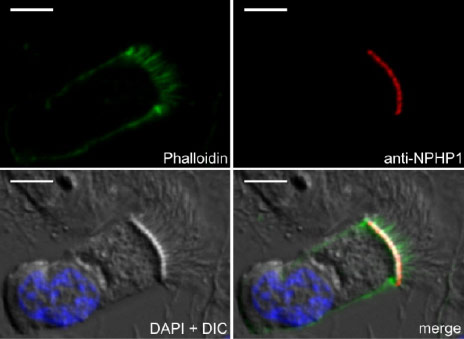
SELECTED PUBLICATIONS
Kieckhofer, E., Slaats, G.G., Ebert, L.K., Albert, M.C., Dafinger, C., Kashkar, H., Benzing, T., and Schermer, B. (2022) Primary cilia suppress Ripk3-mediated necroptosis. Cell Death Discov, 8(1): 477.
Fabretti, F., Tschernoster, N., Erger, F., Hedergott, A., Buescher, A.K., Dafinger, C., Reusch, B., Köntges, V.K., Kohl, S., Bartram, M.P., Weber, L.T., Thiele, H., Altmueller, J., Schermer, B., Beck, B.B., and Habbig, S. (2021) Expanding the spectrum of FAT1 nephropathies by novel mutations that affect Hippo signaling. Kidney International Reports. 6, 1368-1378
Butt, L., Unnersjo-Jess, D., Hohne, M., Edwards, A., Binz-Lotter, J., Reilly, D., Hahnfeldt, R., Ziegler, V., Fremter, K., Rinschen, M.M., Helmstadter, M., Ebert, L.K., Castrop, H., Hackl, M.J., Walz, G., Brinkkoetter, P.T., Liebau, M.C., Tory, K., Hoyer, P.F., Beck, B.B., Brismar, H., Blom, H., Schermer, B.*, and Benzing, T.* (2020) A molecular mechanism explaining albuminuria in kidney disease. Nat Metab, 2(5): 461-474.
Schermer, B., Fabretti, F., Damagnez, M., Di Cristanziano, V., Heger, E., Arjune, S., Tanner, N.A., Imhof, T., Koch, M., Ladha, A., Joung, J., Gootenberg, J.S., Abudayyeh, O.O., Burst, V., Zhang, F., Klein, F., Benzing, T., and Müller, R.-U. (2020) Rapid SARS-CoV-2 testing in primary material based on a novel multiplex RT-LAMP assay. PLOS ONE, 15(11): e0238612.
Muller, R.U. and Schermer, B. (2020) Hippo signaling-a central player in cystic kidney disease? Pediatr Nephrol, 35(7): 1143-1152.
Jain, M., Kaiser, R.W.J., Bohl, K., Hoehne, M., Gobel, H., Bartram, M.P., Habbig, S., Muller, R.U., Fogo, A.B., Benzing, T., Schermer, B., Hopker, K., and Slaats, G.G. (2019) Inactivation of Apoptosis Antagonizing Transcription Factor in tubular epithelial cells induces accumulation of DNA damage and nephronophthisis. Kidney Int, 95(4): 846-858.
Dafinger, C., Rinschen, M.M., Borgal, L., Ehrenberg, C., Basten, S.G., Franke, M., Hohne, M., Rauh, M., Gobel, H., Bloch, W., Wunderlich, F.T., Peters, D.J.M., Tasche, D., Mishra, T., Habbig, S., Dotsch, J., Muller, R.U., Bruning, J.C., Persigehl, T., Giles, R.H., Benzing, T., Schermer, B.*, and Liebau, M.C.* (2018) Targeted deletion of the AAA-ATPase Ruvbl1 in mice disrupts ciliary integrity and causes renal disease and hydrocephalus. Exp Mol Med, 50(6): 75.
Kohli, P., Hohne, M., Jungst, C., Bertsch, S., Ebert, L.K., Schauss, A.C., Benzing, T., Rinschen, M.M., and Schermer, B. (2017) The ciliary membrane-associated proteome reveals actin-binding proteins as key components of cilia. EMBO Rep 18(9): 1521-1535.
Rinschen, M.M., Grahammer, F., Hoppe, A.K., Kohli, P., Hagmann, H., Kretz, O., Bertsch, S., Hohne, M., Gobel, H., Bartram, M.P., Gandhirajan, R.K., Kruger, M., Brinkkoetter, P.T., Huber, T.B., Kann, M., Wickstrom, S.A., Benzing, T., and Schermer, B. (2017) YAP-mediated mechanotransduction determines the podocyte's response to damage. Sci Signal 10(474).
Habbig, S., Bartram, M. P., Muller, R. U., Schwarz, R., Andriopoulos, N., Chen, S., Sagmuller, J. G., Hoehne, M., Burst, V., Liebau, M. C., Reinhardt, H. C., Benzing, T., Schermer, B. (2011) NPHP4, a cilia-associated protein, negatively regulates the Hippo pathway. J Cell Biol 193:633-642.
